Wednesday, August 5, 2009
Pleasant invasions surprise
I am looking forward to more great talks!
Tuesday, August 4, 2009
Species interactions & evolution
I've been in the organized session on species interactions and evolution all morning and there were some great talks (e.g., Silvertown, Ackerly, Cavender-Bares, etc.). But I think what really got me excited were some of the questions after each talk. Following Jonathan Silvertown's talk, Steve Hubbell asked some questions that get to the heart of addressing what phylogenies mean for community assembly. Silvertown showed that within plots, species of a large South African family of plants in the Fynbos seemed to spatially segragate according to hydrological niches and that within these plots there was a lack of phylogenetic signal in this niche. Hubbell then asked two critical questions: How many other species (in other families) co-occur in these niches and if related species have similar niches at a larger scale. To me this is at the core of uderstanding how phylogenies inform our understanding of community assembly. Basically, what haven't we measured? If we include all sister species into a phylogeny, do we change our understanding of the processes structuring communities?
More later!
Monday, July 27, 2009
Incommunicado Apologies & ESA next week
Sorry for the lack of postings as of late. I've just started a professorship, which involved a move from my postdoc in California to my new position in Toronto, Canada, and I guess I failed to completely appreciate how much of a time sink all this would be.
Anyway, I've resurfaced and will be blogging from the ESA annual meeting in Albuquerque next week. I'll be speaking in a workshop on Sunday that deals with whether exotics are different from native species, and there are some great ecologists that will be speaking (like Dov Sax, John Maron, Dave Richardson, Peter Kotanen and John D. Parker). Also, I'm talking in an organized oral session on species interactions and relatedness with a stellar group (e.g., Jeannine Cavender-Bares, Jonathan Silvertown, David Ackerly, Steve Kembel, Jonathan Davies and Andras Prinzing), which is a little daunting. Looking at the schedule, there are way too many interesting talks and my schedule is already double-booked with talks I want to attend. Rock, Paper, Scissors. Should be great time!
Hope to see you in Albuquerque,
Marc
Thursday, June 11, 2009
The sushi of tomorrow… Jellyfish rolls?
In recent years, jellyfish outbreaks have become more frequent and more severe. These outbreaks can have lasting ecological and economic consequences. They can wreak havoc on the tourist industry by closing beaches and harming swimmers, cause power outages by blocking cooling intakes at coastal power plants, reduce commercial fish abundance via competition and predation, spread fish parasites, burst fishing nets, and contaminate catches.
A review by Anthony Richardson and his collaborators suggests that human activities such as overfishing, eutrophication, climate change, translocation, and habitat modification have dramatically increased jellyfish numbers. Their research, which was published this week in Trends in Ecology and Evolution, highlights that the structure of pelagic ecosystems can abruptly transition from one that is dominated by fish to one that is dominated by jellyfish.
Richardson and his collaborators present a potential mechanism to explain how local jellyfish aggregations can spread, displace fish, and form an alternative stable state to fish-dominated ecosystems. Jellyfish are like the opportunistic weed of the sea, giving them an edge in environments stressed by climate change, eutrophication, and overfishing. In these disturbed environments, the abundance of jellyfish relative to filter-feeding fish increases until a tipping point is reached. Under normal conditions, filter-feeding fish keep jellyfish populations in check via competition for planktonic food and (perhaps) predation on an early life-stage of the jellyfish. At the tipping point, jellyfish numbers are such that they begin to overwhelm any control of their vulnerable life-cycle stages by fish predators. At the same time, jellyfish progressively eliminate competitors and predators via their predation on fish eggs and larvae. As jellyfish abundance increases, sexual reproduction becomes more efficient, allowing them to infest new habitats where fish might have formally controlled jellyfish numbers.
Richardson and his collaborators suggest that one way to hit the brakes on what they call the “the never-ending jellyfish joyride” is to harvest more jellyfish for human consumption. Jellyfish have been eaten for more than 1000 years in China, where they are often added to salads. In Japan they are served as sushi and in Thailand they are turned into a crunchy noodle concoction. Although the taste and texture of jellyfish might not be appealing to some westerners, I for one have yet to meet a sushi that I didn’t like. Of course, jellyfish harvesting is unlikely to return systems to their fish-dominated state if the stresses that caused the ecosystem shift remain.
Richardson, A. J., A. Bakun, G. C. Hays, and M. J. Gibbons. 2009. The jellyfish joyride: Causes, consequences and management responses to a more gelatinous future. Trends in Ecology and Evolution, 24 (6), 312-322 DOI: 10.1016/j.tree.2009.01.010
Thursday, May 28, 2009
How long does it take for an ecosystem to recover?
In a recent PLoS ONE paper, Jones and Schmitz attempt to answer this question by reviewing 240 published studies that examine post-disturbance ecosystem diversity and function. While they report that many ecosystems recover on the order of decades and that this is likely more rapid than previously thought, there are some important caveats. First, is that only about half of the 240 studies report a recovered state and either they were not carried out long enough or there are certain types of disturbances or systems where recovery takes much longer. Second is that there are important differences among habitat types. For example benthic algal recovery to hurricanes or oil spills may take 2-10 years, while the recovery of tree diversity to logging may take 20 to 100 years (or more). Thirdly, different measures of ecosystems general resulted in differing recovery times. For example, bird populations may recover quite quickly to logging (likely because they are migratory), whereas soil microbial communities and processes may take many decades due to changes in the soil environment. Finally, the nature of the disturbance can be an important determinant of time to recovery. Logging and agriculture require the greatest recovery time, while large storms and oil spills appear to require relative little time.
While these results may give us a general picture of ecosystem recovery, the data they use highlight the importance of knowing how disturbance type affect recovery and how different ecosystem measures can alter recovery time estimates.
Jones, H., & Schmitz, O. (2009). Rapid Recovery of Damaged Ecosystems PLoS ONE, 4 (5) DOI: 10.1371/journal.pone.0005653
Wednesday, May 20, 2009
Fire and the changing world
Wildfires have been a part of the Earth’s biota likely since organisms first died and dried on land. Ecosystems have been shaped by fire, numerous organisms have evolved strategies to cope with fire and human cultural development has close tied to fire. In a recent review paper in Science by David Bowman, Jennifer Balch and colleagues, they asked the question: how have fires changed and what does the future look like? Human activities are changing fire patterns and climate change may be entering a feedback with fire. Global warming has been linked to increases in extreme fire weather, making large, destructive fires more probable. However, these large fires feedback into this loop because they release compounds that have strong greenhouse effects. Further, smoke plumes inhibit cloud formation, reinforcing the dry conditions that lead to the fires in the first place.
They argue that fire needs to be incorporating into models of climate change and especially those that link ecosystem properties climate change. Fire may change the distribution of specific habitat types beyond that predicting by responses to climate change alone.
Bowman, D., Balch, J., Artaxo, P., Bond, W., Carlson, J., Cochrane, M., D'Antonio, C., DeFries, R., Doyle, J., Harrison, S., Johnston, F., Keeley, J., Krawchuk, M., Kull, C., Marston, J., Moritz, M., Prentice, I., Roos, C., Scott, A., Swetnam, T., van der Werf, G., & Pyne, S. (2009). Fire in the Earth System Science, 324 (5926), 481-484 DOI: 10.1126/science.1163886
Monday, May 18, 2009
It's not me, It's you: self recognition and plant responses to herbivory

Karban, R., & Shiojiri, K. (2009). Self-recognition affects plant communication and defense Ecology Letters, 12 (6), 502-506 DOI: 10.1111/j.1461-0248.2009.01313.x
Tuesday, May 12, 2009
Hurricanes might contribute to global warming
The annual amount of carbon dioxide a forest absorbs from the atmosphere is determined by the ratio of tree growth to tree mortality each year. When hurricanes cause extensive tree mortality, not only are there fewer trees in the forest to absorb greenhouse gases, but these tree die-offs also emit carbon dioxide, thus potentially warming the climate.
Using field measurements, satellite image analyses, and empirical models to evaluate forest and carbon cycle impacts of hurricanes, the researchers established that an average of 97 million trees have been affected each year for the past 150 years over the continental United States, resulting in a 53-million ton annual biomass loss and an average carbon release of 25 million tons per year. Over the period of 1980–1990, released CO2 potentially offset carbon absorption by forest trees by 9–18% over the entire United States. Impacts on forests were primarily located in Gulf Coast areas such as southern Texas, Louisiana, and Florida, but significant impacts also occurred in eastern North Carolina.
These results have important implications for evaluating positive feedback loops between global warming and environmental change.
Zenga, H., J. Q. Chambers, R. I. Negrón-Juárez, G. C. Hurtt, D. B. Baker, and M. D. Powell. (2009). Impacts of tropical cyclones on U.S. forest tree mortality and carbon flux from 1851 to 2000. PNAS, 106 (19), 7888-7892. DOI:10.1073/pnas.0808914106
Wednesday, May 6, 2009
Biological carbon pump potentially slows down with sea surface warming

Biological activity in the world open ocean’s surface is characterized by autotrophic and by heterotrophic processes. Phytoplankton organisms take up dissolved CO2 (dissolved inorganic carbon, DIC) and together with other inorganic nutrients and light they produce biomass (particulate organic carbon, POC) and dissolved organic carbon (DOC). By these processes marine phytoplankton is responsible for approximately half of the worlds primary production. These two carbon compounds (POC and DOC) either sink down to the deep ocean (which is basically the biological carbon pump) or they are consumed by other trophic levels. One important part of the planktonic food web is the microbial community which consists of bacteria (smaller than 3 µm), auto- and heterotrophic flagellates and other protists (larger than three µm). This community takes up both POC and DOC and by respiration recycles these carbon compounds back into DIC. Thus in terms of carbon flux the microbial community potentially competes with the biological carbon pump.
In a mesocosm experiment with natural marine plankton Julia Wohlers and her colleagues manipulated future ocean surface warming and measured the carbon flux during the plankton bloom peak. Whereas in this experiment phytoplankton biomass production (POC of autotrophs) was not affected by warming the authors found that respiration by the microbial community, in particular by organism larger than 3 µm, significantly increased. This increase in respiration led to a significant decrease in net DIC reduction in the whole planktonic foodweb. The results are a potential sign for future declining carbon sequestration by biological processes in the world oceans.
Julia Wohlers, Anja Engel, Eckart Zöllner, Petra Breithaupt, Klaus Jürgens, Hans-Georg Hoppe, Ulrich Sommer and Ulf Riebesell (2009). Changes in biogenic carbon flow in response to sea surface warming. Proceedings of the National Academy of Sciences. DOI:10.1073/pnas.0812743106
Friday, May 1, 2009
Enrichment and diversity loss: a mechanism tested
To paraphrase Thomas Henry Huxley: How stupid of us not to have thought of that!
As reveled in the most recent issue of Science, Hautier et al. performed an exceedingly simple experiment; they added light to the understory of plant communities with or without nitrogen additions. They made two compelling observations. First, when communities were enriched without elevated light, they lost about 3 of the 6 initial species compared to the control, while light addition in the enriched communities maintained the 6 member community (as did a light only treatment). The second result was that the light plus nitrogen treatment obtained much higher biomass than either the nitrogen or light only treatments, and in fact the light only treatment did not significantly increase productivity, meaning that the communities are not normally light-limited. Further, they failed to detect any elevated belowground competition for other resources.
These results reveal that nutrient enrichment causes diversity loss because increased plant size increases light competition and plants that grow taller with elevated nitrogen are better light competitors. An old problem solved with the right experiment.
Hautier, Y., Niklaus, P., & Hector, A. (2009). Competition for Light Causes Plant Biodiversity Loss After Eutrophication Science, 324 (5927), 636-638 DOI: 10.1126/science.1169640
Wednesday, April 22, 2009
People value rare species; at least from their computers

Cleverly, they created a French website where visitors could select to view either a slideshow of common or rare species (and the links randomly changed positions on the site). The trick was that a download status bar appears and freezes near the end, and so Angula and Courchamp were able to measure how many visitors selected the rare species show and how long they waited until they gave up. Visitors were much more likely to select the rare species and to wait longer to see them.
I think that this study is extremely neat for two reasons. First it offers a novel way to quantify valuation, and second, it shows how the internet can be used to assess conservation issues in an efficient low-cost way.
Now will they please just show us the pictures of the cute, endangered species!
Angulo, E., & Courchamp, F. (2009). Rare Species Are Valued Big Time PLoS ONE, 4 (4) DOI: 10.1371/journal.pone.0005215
Friday, April 17, 2009
A mechanism on why communities of exotic species are less diverse than communities of native species
 Plant communities dominated by exotics tend to be less diverse than plant communities dominated by natives. Apparently, few people have been curious enough to plan an experiment to try to further understand why this is the case. A recent paper in ecology letters Brian Wilsey and collaborators showed the results of an experiment designed to explore this. What they did is to create monocultures of a series of exotics and natives species, and mix cultures of exotics (a mix of 9 exotics, zero natives ) and mix cultures of natives (9 natives, zero exotics). They found that large exotics (plants with high aboveground biomass) tended to be even bigger when growing in mix cultures than in the monocultures, so big plants got bigger, which tend to reduce plant richness since it may displace other plants. On the other hand, for natives, small plants tended to get bigger, which is a mechanism for promoting biodiversity (communities may be more even). This research highlights the importance of understanding the mechanisms of plant coexistence and the fact that exotic species may behave very differently than native species.
Plant communities dominated by exotics tend to be less diverse than plant communities dominated by natives. Apparently, few people have been curious enough to plan an experiment to try to further understand why this is the case. A recent paper in ecology letters Brian Wilsey and collaborators showed the results of an experiment designed to explore this. What they did is to create monocultures of a series of exotics and natives species, and mix cultures of exotics (a mix of 9 exotics, zero natives ) and mix cultures of natives (9 natives, zero exotics). They found that large exotics (plants with high aboveground biomass) tended to be even bigger when growing in mix cultures than in the monocultures, so big plants got bigger, which tend to reduce plant richness since it may displace other plants. On the other hand, for natives, small plants tended to get bigger, which is a mechanism for promoting biodiversity (communities may be more even). This research highlights the importance of understanding the mechanisms of plant coexistence and the fact that exotic species may behave very differently than native species.Wilsey, B., Teaschner, T., Daneshgar, P., Isbell, F., & Polley, H. (2009). Biodiversity maintenance mechanisms differ between native and novel exotic-dominated communities Ecology Letters, 12 (5), 432-442 DOI: 10.1111/j.1461-0248.2009.01298.x
Sunday, April 5, 2009
Climate change increases West Nile Virus outbreaks in the U.S.
In the study, Dr. Jonathan Soverow and his collaborators matched more than 16,000 confirmed West Nile cases in 17 states to local meteorological data.
Warmer temperatures had the greatest effect on outbreaks. By extending the length of the mosquito breeding season and decreasing the amount of time it takes mosquitoes to reach their adult, biting stage, warmer weather means more biting mosquitoes longer. Moreover, increasing temperature speeds multiplication of the virus within insects, so mosquitoes in warmer climates have a greater viral load, making them more likely to infect humans.
Increased precipitation was also correlated with higher rates of West Nile Virus infection. A single, heavy rainstorm resulting in two or more inches of rain increased infection rates by 33%, while smaller storms had less of an effect on infection rates. Heavier rainfall events can increase disease prevalence by creating pools of water in which mosquitoes can breed and by increasing humidity, which stimulates mosquitoes to bite and breed. Total weekly rainfall had a smaller but significant effect on West Nile Virus infections, with an increase of 0.75 inch of rain/week increasing the number of infections by about 5%.
Warmer, wetter weather patterns might expand the niches of the mosquito species that carry West Nile Virus. In California, for instance, several mosquito species carrying the West Nile Virus have extended their ranges into higher elevations and coastal areas as temperatures have warmed. Changing weather patterns might also affect certain species of birds that are reservoirs for West Nile Virus. For example, droughts can push bird populations into urban areas, making West Nile Virus outbreaks in human populations more likely.
Soverow, J.E., G.A. Wellenius, D.N. Fisman, and M.A. Mittleman. 2009. Infectious disease in a warming world: How weather influenced West Nile Virus in the United States (2001-2005). Environmental Health Perspectives. Online 16 March 2009 DOI: 10.1289/ehp.0800487
Thursday, April 2, 2009
Letting out your little Monet
Here are some examples of artistically augmented publication figures -but if you have other good examples, let me know and I'll add them:

This is from a recent Ecology Letters from Crutsinger, Cadotte (me) and Sanders (2009), 12: 285-292, trying to explain how we partitioned arthropod diversity into spatial components.

This one is from Ellwood et al. (2009) in Ecology Letters 12: 277-284, which shows co-occurrence null histograms for patterns of arthropods at various hight locations on trees.
 This one is from Crutsinger et al (2006) Science 313: 966-968 that displays patterns at differing trophic levels by juxtaposing photos of specific tropic members.
This one is from Crutsinger et al (2006) Science 313: 966-968 that displays patterns at differing trophic levels by juxtaposing photos of specific tropic members.

Finally, the use of drawings and images to illustrate phylogenetic trends in phenotypic evolution is particularly useful. Above are two examples, on the left is from Carlson et al. 2009 Evolution 63: 767-778, showing patterns of darter evolution; and on the right is from Oakley and Cunningham 2002 PNAS 99: 1426-1430, showing evolutionary pathways of compound eyes.

And here's one from Dolph Schluter (2000) American Naturalist 156: S4-S16, using drawings to illustrate how fish morphology corresponds to an abstracted index on the bottom axis.
 Here are two from Joe Baily while working in Tom Whitham's Cottonwood Ecology Group that are effective ways to remind the reader what the treatments or dependent variables were (elk herbivory, leaf shape/genotype) and what the response variables were (bird predation, wood consumption by beavers). The left hand figure is from Baily & Whitham (2003) Oikos 101: 127-134 and the one on the right is from Baily et al. (2004) Ecology 85: 603-608.
Here are two from Joe Baily while working in Tom Whitham's Cottonwood Ecology Group that are effective ways to remind the reader what the treatments or dependent variables were (elk herbivory, leaf shape/genotype) and what the response variables were (bird predation, wood consumption by beavers). The left hand figure is from Baily & Whitham (2003) Oikos 101: 127-134 and the one on the right is from Baily et al. (2004) Ecology 85: 603-608.Here is a great one posted by Ethan on Jabberwocky Ecology on Hurlbert's Unicorn!
Friday, March 27, 2009
The evolutionary meaning of autumn colors

1) Sunscreen: Pigments provide photoprotection against photooxidation during the recovery of nutrients.
2) Leaf warming: Colors absorb light and warm the leaves during cooling temperatures.
3) Coevolution: Tells overwintering insects that the tree is not suitable (poisonous or low nutrition) for hibernation.
4) Camouflage: Many insects lack red photoreceptor, making leaves difficult to see -thus protecting trees from overwintering pests.
5) Unpalatability: Pigments (e.g., red -anthocyanins) are unpalatable.
So, we may quibble about particular hypotheses, but the point for me is that there may be deeper explanations as to why certain species produce the vivid colors they do. At a minimum, Archetti provides ammunition to experimental botanists and evolutionary biologists for testing new hypotheses. I'll never look at an autumn forest the same again.
Archetti, M. (2009). Classification of hypotheses on the evolution of autumn colours Oikos, 118 (3), 328-333 DOI: 10.1111/j.1600-0706.2008.17164.x
Archetti, M. (2008). Phylogenetic analysis reveals a scattered distribution of autumn colours Annals of Botany, 103 (5), 703-713 DOI: 10.1093/aob/mcn259
Archetti, M., Döring, T., Hagen, S., Hughes, N., Leather, S., Lee, D., Lev-Yadun, S., Manetas, Y., Ougham, H., & Schaberg, P. (2009). Unravelling the evolution of autumn colours: an interdisciplinary approach Trends in Ecology & Evolution, 24 (3), 166-173 DOI: 10.1016/j.tree.2008.10.006
Monday, March 23, 2009
Conserve now or wait for the data?
The philosophical divide between protect now-learn later versus the need for detailed information to maximize resources appears bridgeable. It seems that by just accumulating some rough data may go a long way towards making those important conservation decisions. Of course, the irony is that this study needed 20 years of data to adequately assess this.
Grantham, H., Wilson, K., Moilanen, A., Rebelo, T., & Possingham, H. (2009). Delaying conservation actions for improved knowledge: how long should we wait? Ecology Letters, 12 (4), 293-301 DOI: 10.1111/j.1461-0248.2009.01287.x
Tuesday, March 17, 2009
Being a clover isn’t always so lucky
I always think of Trifolium species as being particularly common and widely distributed, but there are some that are threatened and potentially tell us about the threats faced by imperiled plant populations. In fact, while a number of North American Trifolium species have successfully invaded North America, but T. montanum is not, according to the USDA Plants Database. These results reveal that these negative effects affect plants at different stages of their life cycle (growth to maturity vs. recuitment) and that log-term persistence of these populations requires management activities that ameliorate both of these effects.
SCHLEUNING, M., NIGGEMANN, M., BECKER, U., & MATTHIES, D. (2009). Negative effects of habitat degradation and fragmentation on the declining grassland plant Trifolium montanum Basic and Applied Ecology, 10 (1), 61-69 DOI: 10.1016/j.baae.2007.12.002
Monday, March 16, 2009
A roadmap to generalized linear mixed models
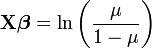 In a recent paper in TREE, Ben Bolker (from the University of Florida) and colleagues describe the use of generalized linear mixed models for ecology and evolution. GLMMs are used more and more in evolution and ecology given how powerful they are, basically because they allow the use of random and fix effects and can analyze non-normal data better than other models. The authors made a really good job at explaining what to use when. Despite the fact that you need more than basic knowledge of stats to fully understand this guide, I think that people should take a look at it before starting to plan their projects, since it outlines really well all the possible alternatives (and challenges) that one can have when analyzing data. This article also describes what is available in each software package; this is really useful since is not obvious with program in SAS or R you need to use when dealing with some specific GLMMs.
In a recent paper in TREE, Ben Bolker (from the University of Florida) and colleagues describe the use of generalized linear mixed models for ecology and evolution. GLMMs are used more and more in evolution and ecology given how powerful they are, basically because they allow the use of random and fix effects and can analyze non-normal data better than other models. The authors made a really good job at explaining what to use when. Despite the fact that you need more than basic knowledge of stats to fully understand this guide, I think that people should take a look at it before starting to plan their projects, since it outlines really well all the possible alternatives (and challenges) that one can have when analyzing data. This article also describes what is available in each software package; this is really useful since is not obvious with program in SAS or R you need to use when dealing with some specific GLMMs.Bolker, B., Brooks, M., Clark, C., Geange, S., Poulsen, J., Stevens, M., & White, J. (2009). Generalized linear mixed models: a practical guide for ecology and evolution Trends in Ecology & Evolution, 24 (3), 127-135 DOI: 10.1016/j.tree.2008.10.008
Friday, March 6, 2009
Salamaders and climate change -impending extinctions?
The authors attempted to determine the cause of these declines and found that neither habitat loss or the chytridiomycosis fungal disease implicated in other declines explained these salamander declines. The authors hypothesize that these declines are a direct result of climate change –namely changing temperature and humidity. If so, we may be witnessing some of the first extinctions that are directly caused by climate change.
S. M. Rovito, G. Parra-Olea, C. R. Vasquez-Almazan, T. J. Papenfuss, D. B. Wake (2009). Dramatic declines in neotropical salamander populations are an important part of the global amphibian crisis Proceedings of the National Academy of Sciences, 106 (9), 3231-3236 DOI: 10.1073/pnas.0813051106
Post script:
We had a comment questioning the use of climate change as an explanation and here is my response.
Science works by finding parsimonious explanations, until through experimentation or observation another, better explanation emerges. The previous explanations of habitat loss or fungal infections were not supported. These habitats, known as cloud forests, are very humid. The lungless salamanders have no lungs and instead breath through their skin, which must be kept moist. These forest are becoming drier, hence the probable connection. Here's a quote from the paper:
"Pounds et al. (25) used modeling to show that largescale warming led to a greater decrease in relative humidity at Monteverde compared to that caused by deforestation. Species of cloud forest salamanders that can still be found rely at least
in part on bromeliads. Bromeliads depend on cloud water deposition and are predicted to be articularly vulnerable to climate change (26, 27)."
Doesn't sound like "magic" to me, rather a robust hypothesized mechanism worthy of more testing. Given that species are going extinct, it is important to suggest likely mechanisms, providing an impetus for more research.
For those that think that scientists use climate change as boogey man to scare up more research funding (i.e., Crichton), please read the science. You'll discover honest, hardworking folks that are trying to understand this changing world and whose research can only benefit you , me and the salamanders.
Sunday, March 1, 2009
Phytoplankton motility and morphology might influence red tides
Although thin layer formation was previously thought to be solely influenced by abiotic forces, a recent paper in Science by William M. Durham and colleagues suggests that plankton’s swimming and shape play a role. Many phytoplankton species swim upward against gravity. When the water is calm, they swim up in a straight path. But add ocean currents to the equation, and the plankton start to encounter vertical shear where layers of faster- and slower- moving water meet. These shear forces can cause the plankton to tumble and spin instead of swimming straight up. The tumbling plankton become trapped in these regions of high shear, accumulating in a thin layer. The strength of the shear forces interacts with the morphology of the plankton to determine which species get trapped. For instance, bottom heavy species require higher shear to knock them off their straight path. Durham et al.’s findings suggest that vertical shear and cell morphology could be important predictors of red tides.
W. M. Durham, J. O. Kessler, R. Stocker (2009). Disruption of Vertical Motility by Shear Triggers Formation of Thin Phytoplankton Layers Science, 323 (5917), 1067-1070 DOI: 10.1126/science.1167334
Friday, February 27, 2009
Peace and conservation biology

These news are really sad, and I hope that we can live in peace for the benefits of all the species living in here (including, of course, Homo sapiens).
THOR HANSON, THOMAS M. BROOKS, GUSTAVO A. B. DA FONSECA, MICHAEL HOFFMANN, JOHN F. LAMOREUX, GARY MACHLIS, CRISTINA G. MITTERMEIER, RUSSELL A. MITTERMEIER, JOHN D. PILGRIM (2009). Warfare in Biodiversity Hotspots Conservation Biology DOI: 10.1111/j.1523-1739.2009.01166.x
Sunday, February 22, 2009
The incredible spreadable weeds
Curtis C. Daehler (2009). Short Lag Times for Invasive Tropical Plants: Evidence from Experimental Plantings in Hawai'i PLoS ONE, 4 (2) DOI: 10.1371/Journal.pone.0004462
Friday, February 20, 2009
Increased access to science, but who gets to publish?
J. A. Evans, J. Reimer (2009). Open Access and Global Participation in Science Science, 323 (5917), 1025-1025 DOI: 10.1126/science.1154562
Wednesday, February 18, 2009
Functional traits and trade-offs explain phytoplankton community structure

After attending the presentation by Elena Litchman at the ASLO Aquatic Science Meeting in Nice three weeks ago I came across this paper. Although it was published already two years ago, this works need to be highlighted! Marine phytoplankton is important. It contributes approximately 50% to world primary productivity. Among other factors phytoplankton communities are structured by competition for limiting nutrients (mainly for nitrate and ammonia) in the ocean. Litchman et al. base their paper on the presumption that phytoplankton organisms can achieve higher competitive ability (Tilman’s R*) by different strategies. That is, the organisms can either increase their maximum nutrient uptake and/or growth rate or they decrease the minimum cell quota, the half saturation constant for nutrient uptake and/or their mortality. Litchman et al. tested if they can find constraints and trade-offs on the evolution of better competitive abilities (lower R*) in major phytoplankton groups. Specifically they asked if there is a positive relationship between maximum growth rate and R* which would show a gleaner-opportunist trade-off.
The authors show positive relationships between measurements for growth and nitrate uptake which can constrain the evolution on competitive ability. Indeed major groups of phytoplankton group along these trade-off curves. Whereas coccolithophores e.g. show low nitrate uptake rates and low half-saturation constants, diatoms and dinoflagelates show the opposite nitrate uptake strategy with high uptake rates and high half-saturation constants. A gleaner-opportunist trade-off, i.e. a positive correlation between maximum growth rates and R*which would result in a super species, could not be found across major groups but within the diatoms. The paper gives more results about trait differences among taxonomic groups and allometric scaling relationships. Trade-offs and different strategies in nutrient uptake are discussed in a very concise way either from a mechanistic physiological view as well as from the evolutionary history perspective.
Elena Litchman, Christopher A. Klausmeier, Oscar M. Schofield and Paul G. Falkowski (2009) The role of functional traits and trade-offs in structuring phytoplankton communities: scaling from cellular to ecosystem level. Ecology Letters. DOI: 10.1111/j.1461-0248.2007.01117.x
Sunday, February 15, 2009
The next generation of invasive plants
 The study of plant invasions has usually focused on plants adapted to colonizing areas immediately after a disturbance (early successional species or “weeds” by some definitions). An example of this, is the seminal work of Herbert Baker, with his list of traits to explain why some species become weeds and others do not (e.g. fast growth, production of lots of seeds, good dispersal.). So why do late-successional species (for example, forest understory species) not invade? The answer is: we did not look close enough - they indeed invade!! This is the point of a paper by Patrick Martin and collaborators, who study forests – which are usually known as “invasion resistant systems” – and the colonization of exotic shade-tolerant species in them. Their central point is that there have been larger numbers of introductions of early successional species compared to late successional species (or shade tolerant), and that forest dynamics are much slower than other systems (e.g. a forest gap must be created for a species that need disturbances to colonize). And it is for these reasons that we associate invasive plants solely with early successional species, and we see forests as invasion-resistant systems. We are now observing many highly invasive plants that are not disturbance dependent. These may be a lot harder to control and could have important detrimental effects on native communities.
The study of plant invasions has usually focused on plants adapted to colonizing areas immediately after a disturbance (early successional species or “weeds” by some definitions). An example of this, is the seminal work of Herbert Baker, with his list of traits to explain why some species become weeds and others do not (e.g. fast growth, production of lots of seeds, good dispersal.). So why do late-successional species (for example, forest understory species) not invade? The answer is: we did not look close enough - they indeed invade!! This is the point of a paper by Patrick Martin and collaborators, who study forests – which are usually known as “invasion resistant systems” – and the colonization of exotic shade-tolerant species in them. Their central point is that there have been larger numbers of introductions of early successional species compared to late successional species (or shade tolerant), and that forest dynamics are much slower than other systems (e.g. a forest gap must be created for a species that need disturbances to colonize). And it is for these reasons that we associate invasive plants solely with early successional species, and we see forests as invasion-resistant systems. We are now observing many highly invasive plants that are not disturbance dependent. These may be a lot harder to control and could have important detrimental effects on native communities. Friday, February 13, 2009
40% believe in evolution, but only 25% do not!
However, several reports and blogs about this poll disparage Americans for their lack of scientific sophistication, but I think that the results are far more positive then I would have guessed. Only 25% outright deny evolution! I would have thought a clear majority would take this stance as was shown in 2005. A further 36% do not have an opinion, and as scientists and educators, these folks are the reason why we educate and hold events like Darwin Day. Thank you to all those who work so tirelessly promoting science education and literacy, like those at NCSE.
Wednesday, February 11, 2009
Charles Darwin, founder of evolution AND ecology
Darwin is rightfully seen (or vilified in some quarters) as the founder of modern evolutionary biology. He gave the naturalists of that era an observable and testable mechanism explaining species change and for understanding the similarities and differences among species. As we celebrate Darwin’s 200th birthday and the 150th anniversary of the publication of the Origin of the Species, it seemed right to think about Darwin’s contributions beyond just evolutionary change, namely ecological patterns and processes.
I’ve read Origin probably half a dozen times now and as an ecologist, I am always amazed by the depth and breadth of Darwin’s insights. Every time I read it, there are passages that directly relate to what I happen to be thinking about or working on at the time, which leads me to the conclusion that he thought a lot about what scientists would come to call ecology. Though the word “ecology” wouldn’t be invented for another seven years (by Ernst Haeckel in 1866) and the first ecology text book didn’t appear until 1895 (by Eugenius Warming, and which includes interesting Lamarckian invocations in the last chapter), Darwin thought and wrote about ecology extensively.
In the Origin (1st edition), Darwin makes predictions about ecological patterns. On page 109, he states, “a … larger number of the very common and much diffused or dominant species will be found on the side of larger genera”. That is community dominance likely relays on inherited traits linked to species success. This certainly sounds like the result of some recent, interesting papers (e.g., Strauss et al.*).
Almost the whole discussion in the Struggle for Existence chapter is about ecological interactions and the severity of negative interactions, which stems from the fact that populations, if unchecked, will increase exponentially (i.e., page 116). We all know from work by ecologists such as Connell and Huston that those negative, deterministic interactions can be overridden by non-equilibrium processes, especially disturbances. Here again Darwin’s observations lead him to this conclusion; “If turf which has long been mown …be let to grow, the more vigorous plants gradually kill the less vigorous” and he observes that diversity in a plot goes from 20 species to 11 when the disturbance is removed.
Further, we often think of Darwin’s view of the environment as a selective pressure (e.g., fur thickness), but he also saw the environment as a determinant of species interactions. Lush places support a lot of species and the control of populations is due to competitive interactions, whereas in harsh places, populations are controlled by “injurious action” of the environment (e.g., page 121). Thus there is a shift from biotic to abiotic controls on ecological processes.
I think that we have collectively forgotten that evolution directly informs our expectations and predictions of ecological patterns and processes. While ecological geneticists drove much of the modern synthesis in the mid 1900’s by incorporating ecology (namely selection) into evolutionary processes, the reverse, bringing evolution into ecology is only now really starting to happen. Lets hope this second modern synthesis completes Darwin’s vision.
Tuesday, February 10, 2009
Stability begets diversity
This conundrum has been solved by Ana Carnaval, a postdoctoral researcher in Craig Moritz's lab at UC Berkeley, and colleagues. They use patterns of diversity to identify probable refuges and then support several independent hypotheses about refuge effects on patterns of frog diversity. They show 1) that there is higher genetic diversity within and among refuge populations relative to non-refuges. 2) They show a multi-species pattern of recent population expansion in non-refuges from adjacent refuges. 3) The absence of isolating divergence in non-refuges because of a lack of time. Finally, 4) strong phylogenetic patterns of between refuge structure, indicating periods of isolation and divergence.
This paper reveals that hypotheses about the origin of species diversity in hotspots can be tested by using genetic divergence below the species level. Not only does this strongly support the spatial refuge hypothesis for tropical diversity patterns but it also elegantly intertwines microevolutionary processes with macroevolutionary patterns. There couldn't have been a more appropriate study published in the week before Darwin's birthday.
A. C. Carnaval, M. J. Hickerson, C. F. B. Haddad, M. T. Rodrigues, C. Moritz (2009). Stability Predicts Genetic Diversity in the Brazilian Atlantic Forest Hotspot Science, 323 (5915), 785-789 DOI: 10.1126/science.1166955Sunday, February 8, 2009
Shortening the R curve
 I am a strong proponent of R for all data management, analysis and visualization. It is a truly egalitarian analysis package -open source and community-contributed analysis packages. The true power comes from complete control and automization of your analyzes as well as publicly accessible new functions created by members of the community. However, the drawback for a lot of people has been the rather steep learning curve, as with any programing language. But there are now a plethora of good books available that help shorten this curve. The Human Landscapes blog as reviewed and ranked introductory and reference R books, which should serve as an invaluable resource for those striving to become aRgonauts.
I am a strong proponent of R for all data management, analysis and visualization. It is a truly egalitarian analysis package -open source and community-contributed analysis packages. The true power comes from complete control and automization of your analyzes as well as publicly accessible new functions created by members of the community. However, the drawback for a lot of people has been the rather steep learning curve, as with any programing language. But there are now a plethora of good books available that help shorten this curve. The Human Landscapes blog as reviewed and ranked introductory and reference R books, which should serve as an invaluable resource for those striving to become aRgonauts.
Friday, February 6, 2009
Don’t miss the mechanism when testing for biodiversity effects
John J. Stachowicz, Rebecca J. Best, Matthew E. S. Bracken, Michael H. Graham (2008). Complementarity in marine biodiversity manipulations: Reconciling divergent evidence from field and mesocosm experiments. Proceedings of the National Academy of Sciences DOI: 10.1073/pnas.0806425105
Tuesday, February 3, 2009
Local extinctions reveal metacommunity dynamics.
Kevin C. Burns, Christopher J. Neufeld (2009). Plant extinction dynamics in an insular metacommunity Oikos, 118 (2), 191-198 DOI: 10.1111/j.1600-0706.2008.16816.x
Monday, February 2, 2009
I have one of the worst jobs in science!
 According to Popular Science's annual ranking of the worst jobs in science, I (no really me!) have one of the worst jobs. They list scientists doing triage -that is having to evaluate which species to save given that we can't save all, as being particularly crummy. They specifically cite my study of phylogenetic uniqueness and ecosystem function as an example. Well I guess it is a little depressing to try to evaluate which species should be saved over others, but I don't think it is as bad as a medical waste burner...
According to Popular Science's annual ranking of the worst jobs in science, I (no really me!) have one of the worst jobs. They list scientists doing triage -that is having to evaluate which species to save given that we can't save all, as being particularly crummy. They specifically cite my study of phylogenetic uniqueness and ecosystem function as an example. Well I guess it is a little depressing to try to evaluate which species should be saved over others, but I don't think it is as bad as a medical waste burner...
Tuesday, January 27, 2009
Small experimental plots predict entire ecosystem responses! (if you work in peatlands…)
Magdalena M. Wiedermann, Urban Gunnarsson, Mats B. Nilsson, Annika Nordin, Lars Ericson (2009). Can small-scale experiments predict ecosystem responses? An example from peatlands Oikos DOI: 10.1111/j.1600-0706.2008.17129.x
